Revolutionizing Skin Filling Science
KYLYS AESTHETICS SA
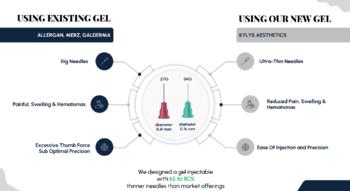
KYLYS AESTHETICS offers a biocompatible hyaluronan-based and thermosensitive dermal filler that has the ability to form microgel structures in the tissue, resulting in a long-lasting effect and no migration from the injection site. Our patented technology allows for injection with an ultra-thin 34G needle, 80% thinner than those used by market alternative, resulting in greater precision, less pain for the patient, and fewer hematomas. With our cutting-edge technology and commitment to research and development, we aim to revolutionize the market of skin filling.
We recently completed all preclinical testing, including biocompatibility, and are now preparing for our 1st pilot clinical trial.
News
From trials to market: medtechs advance clinical programs (startupticker.ch)
KYLYS Aesthetics secures CHF 1 M to advance next-gen hyaluronic acid medical devices (venturelab.swiss)
CHF 1 million to advance KYLYS next-gen hyaluronic acid medical devices (startupticker.ch)
Venture Leaders Biotech scale new heights in Boston (venturelab.swiss)
KYLYS AESTHETICS: The Venture Leader Biotech revolutionizing skin filling science (venturelab.swiss)
The road to Boston: Venture Leaders Biotech 2024 kick-off their roadshow at Swiss Biotech Day (venturelab.swiss)
Ten startups on a business voyage in Boston (startupticker.ch)
Venture Leaders Biotech 2024: 10 innovative startups selected for the Boston roadshow (venturelab.swiss)
FIF supports Fluosphera, Kylys Aesthetics and Consulto (startupticker.ch)
"Serve them, not you" — Interview with Anne Headon of the HUB Entrepreneurship and Innovation UNIL (venturelab.swiss)
Milestones news
Clinical Trial
Biocompatibility testing
Start of stability testing
GMP production